This is the second article in our blog series on corrosion, focusing on the impact corrosive ions can have and which analytical techniques are best-suited to measure corrosive ions. Subscribe below to learn what corrosion is, how it affects the manufacturing process and the damage it can do if not monitored and addressed properly.
 Why Should You Care about Corrosive Ion Measurements?
Why Should You Care about Corrosive Ion Measurements?
Safe and reliable operation of large steam-generating plants depends on the establishment of chemical conditions throughout the steam-water circuit that minimize the corrosion infrastructure materials and suppress the formation of deposits. All primary water consuming industries such as Refining, Chemical, Power Plants, and Pulp & Paper, are affected by corrosion, and each manufacturing step within these industries has a different influence on corrosion.
In dissolved form, both organic and inorganic halides and sulfur compounds accelerate corrosion. The most significant corrosion-causing species are:
- Halides – Inorganic and organic
- Sulfur species - Sulphates & organic sulfur compounds
- Organic acids, inorganic acids, Total Acid (TAN)
- Carbonates & bicarbonates
Corrosion due to sulfur compounds is one of the main challenges in the Refining and Pulp & Paper industries. Even trace levels of inorganic fluoride, chloride, and sulfate present in process water can cause damaging corrosion. The higher the temperature and the pressure of the process water used, the higher the rate of corrosion. Monitoring these compounds is a crucial step in avoiding major corrosion-related costs.
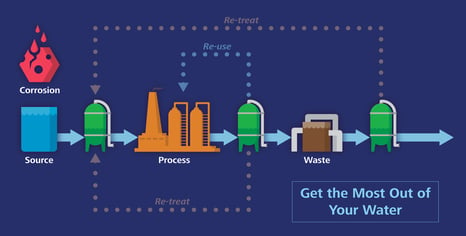 How do corrosive inorganic halides and sulfate impact corrosion rates?
How do corrosive inorganic halides and sulfate impact corrosion rates?
The presence of these corrosive ions in steam is detrimental to the infrastructure used in wet process equipment such as steam generators, heat exchangers, turbine blades, reactor vessel internals, recirculation piping, or connective piping. Minimizing the concentrations of corrosive ions in boiler feed water and the effluent from the condensate polisher reduces potential corrosion damage to various system components. High-purity water is used in all large steam generation industries to reduce corrosion from inorganic halides and sulfates.
Which analytical techniques are best suited for corrosive inorganic ion measurement?
Testing and monitoring corrosive ion concentrations in source water is essential to optimizing the water treatment and proper operation of the demineralization plant. You need to choose the analytical technique(s) based on the source water type and the concentrations of these corrosive ions, as well as the location of the measurements.
Fast and accurate fluoride measurement in source water can be achieved using a fluoride ion selective electrode, as long as the measuring concentrations are in the mg/L range. Chloride & sulfate concentrations from mg/L to g/L are best determined using potentiometric titration. Carbonates & bicarbonates, commonly known as p&m values, are generally determined by potentiometric titration as well. Calcium & magnesium harness and total hardness for evaluating water quality is typically determined by photometric or potentiometric titration.
Integrated hardware and software solutions are best-suited for multiple corrosion ion measurements, including pH and conductivity
Alternatively, High Throughput Ion Chromatography (HPTC) can be used for the simultaneous analysis of corrosive ions. Source water samples containing high destructive ions can be automatically diluted and subsequently filtered before injection by automated inline sample preparation, a versatile technique that saves time and cost by reducing manual sample preparation steps. Furthermore, inline ultrafiltration can be used to protect the column from corrosive source water particles, eliminating the manual sample filtration step and enhancing column longevity.
How do I measure the ppb level of corrosive ions in process water?
Typically, corrosive ions as impurities in low levels are monitored in make-up water, condensate, condensate polisher outlets and, in some cases, feed water. The purity of make-up water is critical in closed water cycles. Monitoring of condensate (usually at the condensate extraction pump discharge) is essential in all plants because it provides the first warning of contaminants that may have resulted from condenser leakage, regenerant chemicals from the makeup plant, or contaminated condensate from a storage system. In plants with condensate polishers, the measurement is performed at the condensate polisher outlet.
Reliable and accurate determination of anions in boiler feed water/steam condensate samples at sub μg/L levels is a challenging analytical problem. When it becomes necessary to identify the sources of contaminants in the steam water circuit that are responsible for increases in conductivity (direct or after cation exchange), the use of ion chromatography can provide particularly valuable information. Furthermore, online measurements of ionic impurities by IC provides useful information regarding the source of contamination and probable rates of corrosion, as well as real time data during the start-up and shut-down of the plants.
We hope that you found this information useful. Subscribe now, so you don't miss the next update on corrosion due to residual moisture. Or go back to our first post on the basics of corrosion.
Posted by Metrohm USA Team
